Metal + dilute acid → Salt + Hydrogen gas
#Reactivity series series#

For example, the result we get out of the reactions between metals and acids, metals and water, and single displacement reactions between metals can be predicted. The reactivity series provides the study of properties and reactivities of the metals, Apart from this reactivity series also provides several other important applications. Highly unreactive May react with some of the strong oxidizing acids May react with some strong oxidizing acids Reacts very slowly with cold water, whereas quickly in boiling water, and very strongly with acids. The Reactivity series of some of the most common metals, arranged in descending order of reactivity: Long Tabular Form of the Reactivity Series Higher ranking metals require greater amounts of energy for their isolation from ores and other compounds.On reaction with dilute HCl or dilute H 2SO 4, all metals that are found above hydrogen in the activity series liberate H 2 gas.The tendency to reduce the metals becomes weaker while traversing down the series.The electro-positivity (tendency to lose electrons) of the elements gets reduced while moving down the reactivity series of metals.These metals can get tarnished or corrode very easily. The reducing tendency of metals at the top of the table has high, that is why they are easily oxidized.Arithmetic Progression - Common difference and Nth term | Class 10 Maths.Area of a Triangle - Coordinate Geometry | Class 10 Maths.Chemical Indicators - Definition, Types, Examples.Class 10 RD Sharma Solutions- Chapter 2 Polynomials - Exercise 2.1 | Set 2.Euclid's Division Algorithm - Real Numbers | Class 10 Maths.Electric Potential and Potential Difference.Step deviation Method for Finding the Mean with Examples.Theorem - The tangent at any point of a circle is perpendicular to the radius through the point of contact - Circles | Class 10 Maths.Section formula – Internal and External Division | Coordinate Geometry.ISRO CS Syllabus for Scientist/Engineer Exam.

#Reactivity series free#
Join Seneca to get 250+ free exam board specfic A Level, GCSE, KS3 & KS2 online courses. Want to learn more about Reactivity Series? Because of this, the metal has to be extracted from the ore (rock) where the metal compound is found. Most metals are only found as compounds because the metal has reacted with other elements in the past. Most metals react with dilute acids to produce a salt and hydrogen gas. Potassium, sodium and lithium all react quickly with cold water to produce a metal hydroxide and hydrogen gas. Platinum, however, is less reactive than copper and so cannot displace copper from a copper sulfate solution. Magnesium + copper sulfate → magnesium sulfate + copper Magnesium is more reactive than copper, so magnesium can displace copper from a copper sulfate solution to create magnesium sulfate. A displacement reaction happens when a more reactive metal (one that forms positive ions more easily) displaces a less reactive metal from a compound.
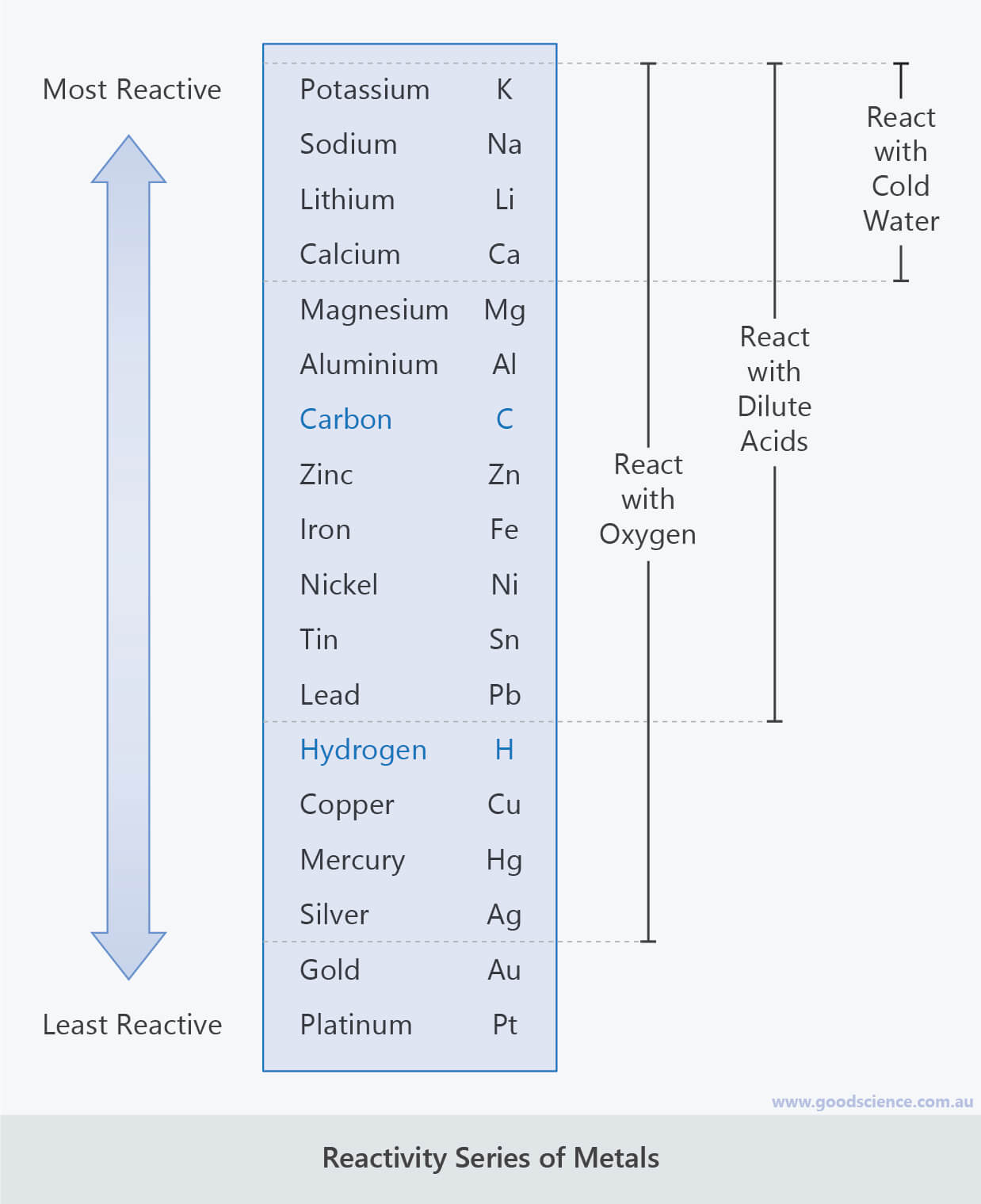
This is called a displacement reaction.Ī metal can only displace another metal from a compound if it is located above it in the reactivity series. These are called native metals.Ī more reactive metal (one that forms positive ions more easily) can displace a less reactive metal from a compound. Very unreactive metals, such as gold and platinum, are found in the Earth’s crust as pure metals. Metals can be arranged in order of their reactivity. The easier it is for a metal to form its positive ion, the more reactive the metal is. When metals react with other substances, the metal atoms always form positive ions.
